PROTEIN FOLDING & ATP
ATP – Beyond Energy
As we know, ATP is the main energy provider in biological systems. ATP, for example, provides the power necessary for muscle contraction, the functioning of neurones or the energy required for maintaining homeostasis. However, ATP fulfils non-energetic roles not less critical to the continuation of life. One of these fundamental roles is the capacity of ATP to increase the thermic stability of protein and promote the correct folding of proteins.
Proteins are chains of amino acids that are elongated by ribosomes in the Endoplasmic Reticulum (ER), based on the sequence of messenger ARN. Although the precise sequence of amino acids is essential to the protein function, its correct tri-dimensional structure is critical for the fulfilment of its biological function. The tri-dimensional structure is dependent on the appropriate protein folding. Protein folding occurs in the reticulum endoplasmic and is known to involve protein chaperones that facilitate the folding often using ATP as energy to promote a specific shape. However, it is only a small aspect of the folding process. Most importantly, ATP induces folding, inhibits aggregation, and increases protein stability post folding1,2.

The Folding Process
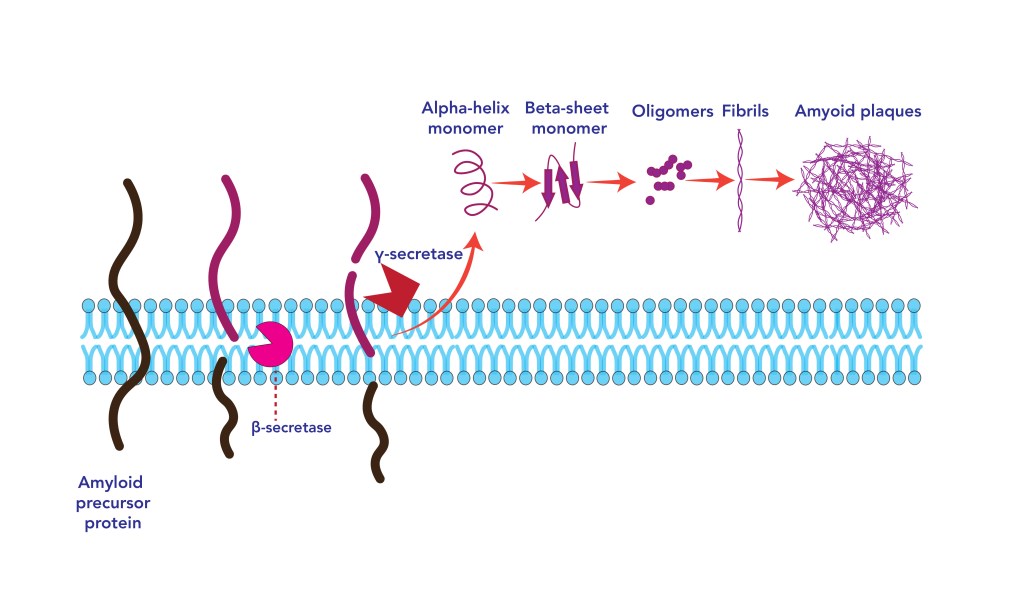
Protein translation and folding occur in the endoplasmic reticulum. First, as mentioned above proteins are translated and elongated from messenger RNA, at this stage they do not yet possess their functional 3D structure. When immature chains expose segments that are prescript to be buried in the folded state, they are prone to accumulate and aggregate with other molecules via hydrophobic patches3. In other words, protein hydrophobic patches have a tendency to associate with each other to avoid contact with the solvent that is hydrophilic, forming dysfunctional protein aggregation that has been associated for example with Alzheimer’s disease and other neurodegenerative diseases. In order to promote correct protein folding and avoid aggregation, protein chaperones are present in the reticulum endoplasmic4 and capture unfolded protein or miss folded protein to enable their correct folding. Some of these chaperones use ATP as a source of energy3 to facilitate this process, others do not5. More recently it was discovered that ATP is also able to facilitate the correct folding of proteins1. Despite all these control systems in place, misfolding does occur. Proteins that fail to fold properly are generally recycled. However, this is not always the case and if a large number of misfolded proteins accumulate in the Reticulum endoplasmic it triggers an ER stress response in the form of the Unfolded Protein Response.
The Unfolded Protein Response
The initial Unfolded Protein Response (UPR) is directed toward reestablishing homeostasis by increasing the expression of chaperone protein and boosting the production of ATP by the mitochondria6. It also enhances the degradation of slowly folding protein7 reducing the folding load of the ER and increasing the degradation of unfolded proteins. The UPR can also boost the size of the ER to increase its folding capacity. On the other extreme the UPR can lead to cell death by apoptosis7. But because misfolding can be caused by an infectious agent, ER stress also leads to the up-regulation of many pro-inflammatory cytokines, including TNFα, IL-1β, IFN-γ, IL-6, and IL-238.
Folding and Autoimmune Disease
Protein translation and folding occur in the endoplasmic reticulum. First, as mentioned above proteins are translated and elongated from messenger RNA, at this stage they do not yet possess their functional 3D structure. When immature chains expose segments that are prescript to be buried in the folded state, they are prone to accumulate and aggregate with other molecules via hydrophobic patches3. In other words, protein hydrophobic patches have a tendency to associate with each other to avoid contact with the solvent that is hydrophilic, forming dysfunctional protein aggregation that has been associated for example with Alzheimer’s disease and other neurodegenerative diseases. In order to promote correct protein folding and avoid aggregation, protein chaperones are present in the reticulum endoplasmic4 and capture unfolded protein or miss folded protein to enable their correct folding. Some of these chaperones use ATP as a source of energy3 to facilitate this process, others do not5. More recently it was discovered that ATP is also able to facilitate the correct folding of proteins1. Despite all these control systems in place, misfolding does occur. Proteins that fail to fold properly are generally recycled. However, this is not always the case and if a large number of misfolded proteins accumulate in the Reticulum endoplasmic it triggers an ER stress response in the form of the Unfolded Protein Response.
Autism – an example:
In recent years some characteristics of the Autism Spectrum Disorder (ASD) have emerged. First, Autism spectrum disorder is associated with neural inflammation (i.e. an elevated production of cytokines including IFN-γ), autoimmunity and autoantibodies13,14. Second, autism spectrum disorder is associated with mitochondrial dysfunction15. Lastly, there is mounting evidence that ASD is caused by protein misfolding16–20. It is interesting to note that mitochondrial dysfunction can be at the same time the cause (not enough ATP production), and the consequence of protein misfolding following UPR. Indeed the stimulation of ATP production by the mitochondria can lead to an overproduction of reactive oxygen species, especially superoxide and trigger mitochondrial dysfunction. Furthermore, ER stress can trigger inflammation and the expression of MHC class II molecules that will promote the autoimmune response.
ER stress and Protein Misfolding have been Identified in Numerous Diseases
Firstly – in neurodegenerative diseases21–23 such as:
- Alzheimer disease.
- Parkinson disease.
- Spinocerebellar ataxia.
- Amyotrophic lateral sclerosis.
- Transmissible spongiform encephalopathies.
- Multiple tauopathies.
- Familial amyloidotic polyneuropathy.
- Fronto temporal dementia.
- Corticobasal degeneration.
- Progressive supra nuclear palsy.
- Dementia with Lewy bodies.
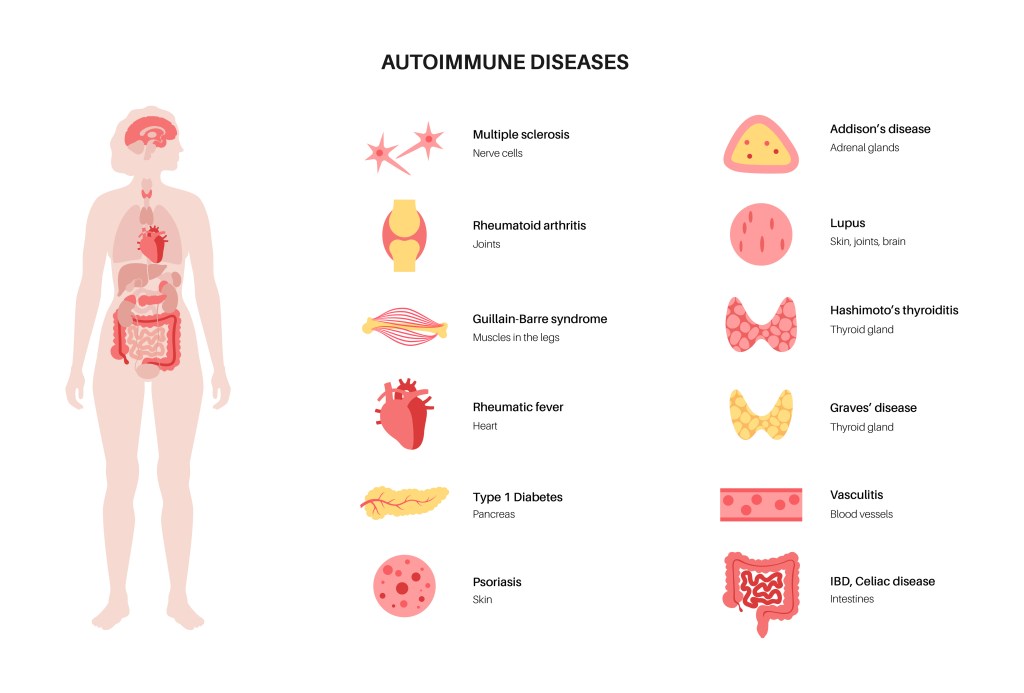
Secondly – in Autoimmune Diseases9–11,24,25 such as:
- inflammatory myopathies
- microscopic polyangiitis
- Rheumatoid arthritis
- Sclerosis
- Celiac Disease
- Crohn’s Disease
- Addison Disease
- Hashimoto Thyroiditis
- Graves Disease
- Dermatomyositis
- Pernicious Anemia
Finally, Protein misfolding and ER stress are involved in both types of diabetes26 and depression27.
Since ER Stress always precedes the development of autoimmune disease, it is expected that the avoidance of ER stress will prevent the development of autoimmune disease and may even resolve it.
Molecular Hydrogen
We know that molecular hydrogen supplementation increases the mitochondrial production of ATP by more than 50% while suppressing the production of superoxide by the mitochondrial complex I (its main producer). Molecular hydrogen supplementation has also demonstrated its anti-inflammatory potential by down regulating NF-kb (the transcription factor responsible for the production of cytosine following the Unfolded Protein Response (UPR).
An increased provision of ATP to the ER is predicted to facilitate correct protein folding by:
- Providing the energy necessary for the correct functioning of the protein chaperone.
- Promoting correct initial folding by itself1.
- Protecting the ER from the aggregation of misfolding protein1 thus decreasing the risk of ER stress.
- Preventing a run-away inflammatory response that increases the risk of developing an autoimmune response.
The action of molecular hydrogen is predicted to significantly suppress the causes leading to ASD as shown in an animal model28, including associated depressive boot since
Reference:
- Kang, J., Lim, L. & Song, J. ATP induces protein folding, inhibits aggregation and antagonizes destabilization by effectively mediating water-protein-ion interactions, the heart of protein folding and aggregation. bioRxiv (2020). 10.1101/2020.06.21.163758
- Ou, X. et al. ATP Can Efficiently Stabilize Protein through a Unique Mechanism. JACS Au 1, 1766-1777 (2021). PMID:34723279;
http://dx.doi.org/10.1021/jacsau.1c00316 - Alaei, L., Ashengroph, M. & Moosavi-Movahedi, A. A. The concept of protein folding/unfolding and its impacts on human health. Adv Protein Chem Struct Biol 126, 227-278 (2021). PMID:34090616;
http://dx.doi.org/10.1016/bs.apcsb.2021.01.007 - Halperin, L., Jung, J. & Michalak, M. The many functions of the endoplasmic reticulum chaperones and folding enzymes. IUBMB Life 66, 318-326 (2014). PMID:24839203;
http://dx.doi.org/10.1002/iub.1272 - Stull, F., Koldewey, P., Humes, J. R., Radford, S. E. & Bardwell, J. C. A. Substrate protein folds while it is bound to the ATP-independent chaperone Spy. Nat Struct Mol Biol 23, 53-58 (2016). PMID:26619265;
http://dx.doi.org/10.1038/nsmb.3133 - Balsa, E. et al. ER and Nutrient Stress Promote Assembly of Respiratory Chain Supercomplexes through the PERK-eIF2α Axis. Mol Cell 74, 877-890.e6 (2019). PMID:31023583;
http://dx.doi.org/10.1016/j.molcel.2019.03.031 - Schröder, M. & Kaufman, R. J. The mammalian unfolded protein response. Annu Rev Biochem 74, 739-789 (2005). PMID:15952902;
http://dx.doi.org/10.1146/annurev.biochem.73.011303.074134 - Bettigole, S. E. & Glimcher, L. H. Endoplasmic reticulum stress in immunity. Annu Rev Immunol 33, 107-138 (2015). PMID:25493331;
http://dx.doi.org/10.1146/annurev-immunol-032414-112116 - Arase, N. & Arase, H. Cellular misfolded proteins rescued from degradation by MHC class II molecules are possible targets for autoimmune diseases. J Biochem 158, 367-372 (2015). PMID:26381536;
http://dx.doi.org/10.1093/jb/mvv093 - Hiwa, R. & Arase, H. Misfolded proteins complexed with MHC class II molecules are targets for autoantibodies. Nihon Rinsho Meneki Gakkai Kaishi 39, 78-83 (2016). PMID:27181239;
http://dx.doi.org/10.2177/jsci.39.78 - Arase, H. Rheumatoid Rescue of Misfolded Cellular Proteins by MHC Class II Molecules: A New Hypothesis for Autoimmune Diseases. Adv Immunol 129, 1-23 (2016). PMID:26791856;
http://dx.doi.org/10.1016/bs.ai.2015.09.005 - Junjappa, R. P., Patil, P., Bhattarai, K. R., Kim, H. R. & Chae, H. J. IRE1α Implications in Endoplasmic Reticulum Stress-Mediated Development and Pathogenesis of Autoimmune Diseases. Front Immunol 9, 1289 (2018). PMID:29928282;
http://dx.doi.org/10.3389/fimmu.2018.01289 - Edmiston, E., Ashwood, P. & Van de Water, J. Autoimmunity, Autoantibodies, and Autism Spectrum Disorder. Biol Psychiatry 81, 383-390 (2017). PMID:28340985;
http://dx.doi.org/10.1016/j.biopsych.2016.08.031 - Onore, C., Careaga, M. & Ashwood, P. The role of immune dysfunction in the pathophysiology of autism. Brain Behav Immun 26, 383-392 (2012). PMID:21906670;
http://dx.doi.org/10.1016/j.bbi.2011.08.007 - Frye, R. E. & Rossignol, D. A. Mitochondrial dysfunction can connect the diverse medical symptoms associated with autism spectrum disorders. Pediatr Res 69, 41R-7R (2011). PMID:21289536;
http://dx.doi.org/10.1203/PDR.0b013e318212f16b - De Jaco, A. et al. A mutation linked with autism reveals a common mechanism of endoplasmic reticulum retention for the alpha,beta-hydrolase fold protein family. J Biol Chem 281, 9667-9676 (2006). PMID:16434405;
http://dx.doi.org/10.1074/jbc.M510262200 - De Jaco, A., Comoletti, D., King, C. C. & Taylor, P. Trafficking of cholinesterases and neuroligins mutant proteins. An association with autism. Chem Biol Interact 175, 349-351 (2008). PMID:18555979;
http://dx.doi.org/10.1016/j.cbi.2008.04.023 - Fujita, E. et al. Autism spectrum disorder is related to endoplasmic reticulum stress induced by mutations in the synaptic cell adhesion molecule, CADM1. Cell Death Dis 1, e47 (2010). PMID:21364653;
http://dx.doi.org/10.1038/cddis.2010.23 - Trobiani, L. et al. The neuroligins and the synaptic pathway in Autism Spectrum Disorder. Neurosci Biobehav Rev 119, 37-51 (2020). PMID:32991906;
http://dx.doi.org/10.1016/j.neubiorev.2020.09.017 - Ulbrich, L. et al. Autism-associated R451C mutation in neuroligin3 leads to activation of the unfolded protein response in a PC12 Tet-On inducible system. Biochem J 473, 423-434 (2016). PMID:26621873;
http://dx.doi.org/10.1042/BJ20150274 - Soto, C. & Estrada, L. D. Protein misfolding and neurodegeneration. Arch Neurol 65, 184-189 (2008). PMID:18268186;
http://dx.doi.org/10.1001/archneurol.2007.56 - Sweeney, P. et al. Protein misfolding in neurodegenerative diseases: implications and strategies. Transl Neurodegener 6, 6 (2017). PMID:28293421;
http://dx.doi.org/10.1186/s40035-017-0077-5 - Soto, C. & Pritzkow, S. Protein misfolding, aggregation, and conformational strains in neurodegenerative diseases. Nat Neurosci 21, 1332-1340 (2018). PMID:30250260;
http://dx.doi.org/10.1038/s41593-018-0235-9 - Hiwa, R. et al. Myeloperoxidase/HLA Class II Complexes Recognized by Autoantibodies in Microscopic Polyangiitis. Arthritis Rheumatol 69, 2069-2080 (2017). PMID:28575531;
http://dx.doi.org/10.1002/art.40170 - Arase, N. et al. Cell surface-expressed Ro52/IgG/HLA-DR complex is targeted by autoantibodies in patients with inflammatory myopathies. J Autoimmun 126, 102774 (2022). PMID:34896887;
http://dx.doi.org/10.1016/j.jaut.2021.102774 - Yamamoto, W. R. et al. Endoplasmic reticulum stress alters ryanodine receptor function in the murine pancreatic β cell. J Biol Chem 294, 168-181 (2019). PMID:30420428;
http://dx.doi.org/10.1074/jbc.RA118.005683 - Ii Timberlake, M. & Dwivedi, Y. Linking unfolded protein response to inflammation and depression: potential pathologic and therapeutic implications. Mol Psychiatry 24, 987-994 (2019). PMID:30214045;
http://dx.doi.org/10.1038/s41380-018-0241-z - Guo, Q. et al. Hydrogen-Rich Water Ameliorates Autistic-Like Behavioral Abnormalities in Valproic Acid-Treated Adolescent Mice Offspring. Front Behav Neurosci 12, 170 (2018). PMID:30127728;
http://dx.doi.org/10.3389/fnbeh.2018.00170 - Satoh, Y. The Potential of Hydrogen for Improving Mental Disorders. Curr Pharm Des 27, 695-702 (2021). PMID:33185151;
http://dx.doi.org/10.2174/1381612826666201113095938 - Zhang, Y. et al. Effects of hydrogen-rich water on depressive-like behavior in mice. Sci Rep 6, 23742 (2016). PMID:27026206;
http://dx.doi.org/10.1038/srep23742
